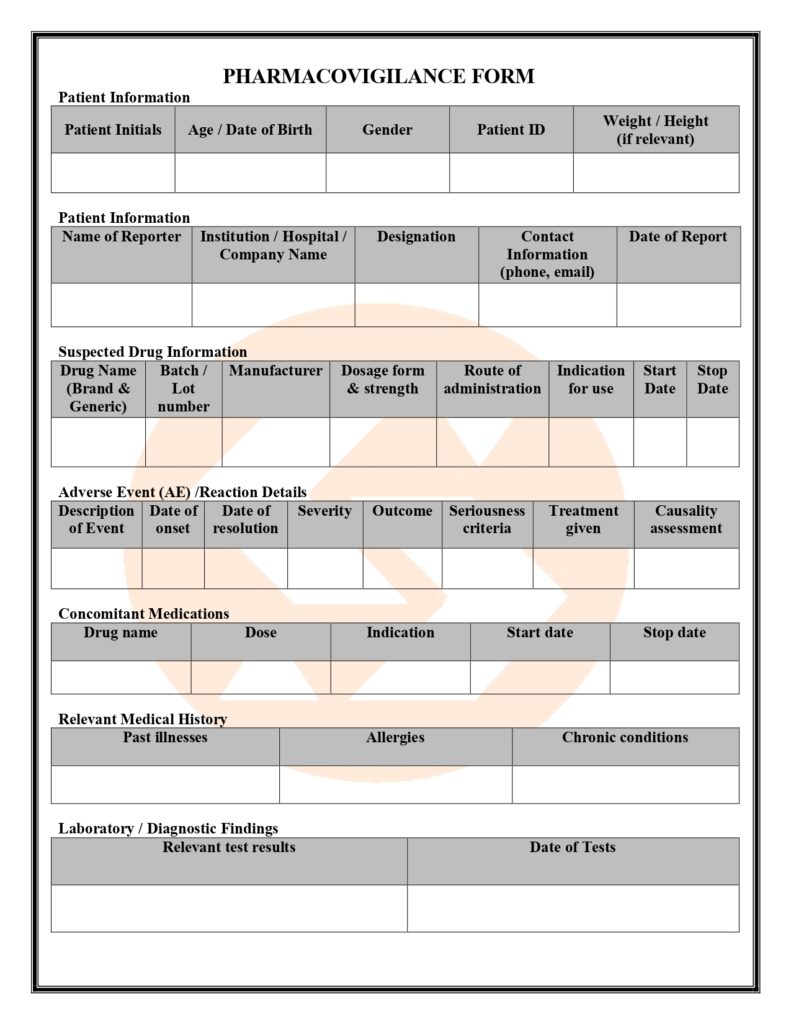
Regulatory & Compliance
Home – Regulatory & Compliance
Licenses & Approvals
Valid GMP Compliance Certificates and Product Registration Authorizations maintained.
ISO Certifications
ISO 9001:2015 & ISO 14001:2015 standards maintained across all departments.
Compliance Statement
Sunshine Pharmaceuticals adheres to WHO, DRAP, and ICH standards — ensuring safety, transparency, and quality at every level.
Regulatory Compliance & Approvals
We operate with full DRAP licensing (DML: 000662) and adhere strictly to WHO, GMP, and ICH regulatory frameworks. Our manufacturing and product registration certificates reflect our commitment to transparency, safety, and global compliance.
Testimonials
Sunshine Pharmaceuticals consistently delivers high-quality and reliable medicines that meet international standards. Their commitment to GMP compliance and affordability truly supports healthcare professionals like me in ensuring better patient outcomes.
